Plugging the Leaks in the Medical Equipment Parts Returns Process with Microsoft Dynamics 365 Field Service
Within the medical device industry, returns can happen on a daily basis. Learn how Microsoft Dynamics 365 Field Service can help you!
Within the medical device industry, returns can happen on a daily basis. Learn how Microsoft Dynamics 365 Field Service can help you!
Table of Content
Are you a medical device company that is losing track of customer returns? Do you want to keep record of the destination of the returned items? Is your product inventory lacking precision due to returns being swept under the rug? Are you losing out on credits owed to you by vendors? If you answered yes to any of those questions, keep reading to see how out-of-the-box Microsoft Dynamics 365 Field Service can help you!
Within the medical device industry, returns can happen on a daily basis. An organization might order a magnet for a work order that is never used and needs to be returned. Secondly, an organization might accept a defective compressor to be repaired and sent back to the main warehouse to be used again in the future. Thirdly, an organization might need to collect a medical device from a customer location that is part of an ending lease that is not being renewed and therefore, they need to accept the equipment back.
Returns can come in many shapes and sizes, and some would say no return is the same. With cloud-based field service automation software, you can empower your parts department to keep track of returns like never before.
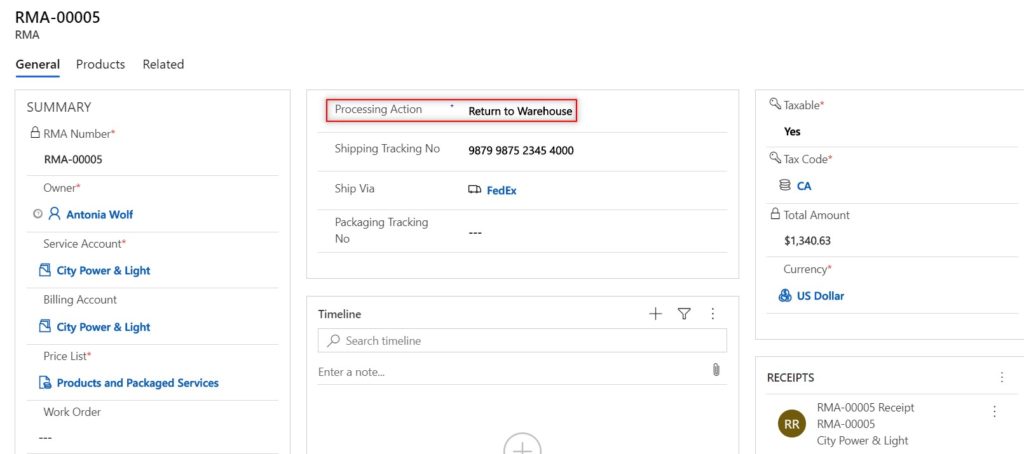
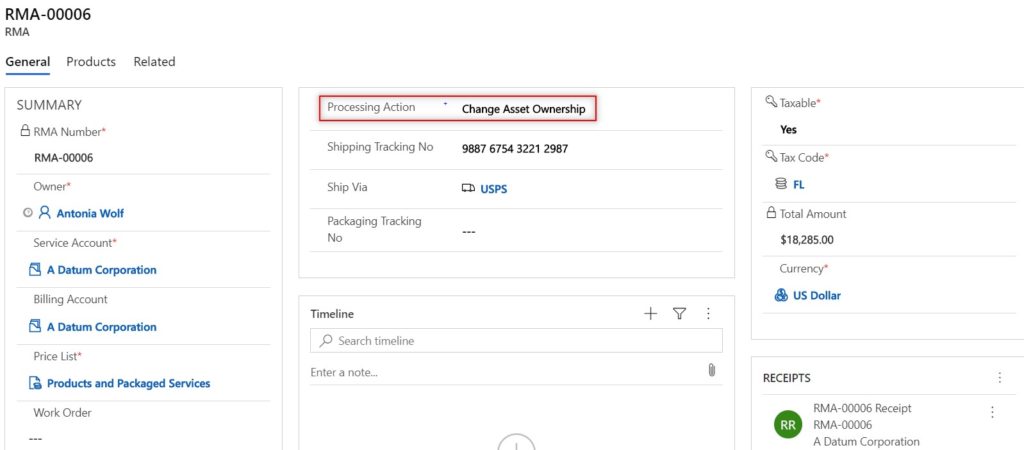
All returns start with an RMA (Return Merchandise Authorization). The RMA can be given a processing action, which notates the type of return and how the return will be processed. From an RMA, you can return the parts back to the OEM, return the parts back to a specific warehouse, or change the equipment ownership. This processing action can be set at the header level to ensure all RMA products follow the same action; however you can also set the processing action for each individual RMA product, giving you freedom to track the different types of return scenarios you may have on a single RMA.
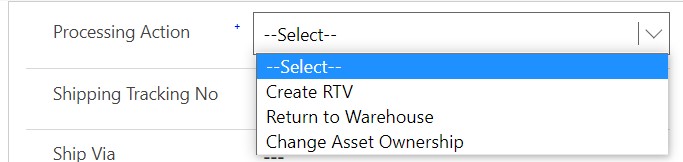
When a customer calls in wanting to return a product, the first step is to create an RMA to track the information on the return, like the customer who is returning the product, the return date, the shipping information, the product(s) and quantities to be returned, if there is an associated work order, the processing action, etc. Depending on the return policy, the RMA might require approval. With Field Service automation software from Microsoft, you can track the reason for a return, decide if the products will be returned to the OEM or to a warehouse, if a credit to the customer is due, and the overall approval process on an RMA. Once the product(s) are delivered to the warehouse, the parts department can review and inspect the returned product(s) to ensure they can be appropriately returned. They can then create an RMA receipt to identify the product(s) were returned.
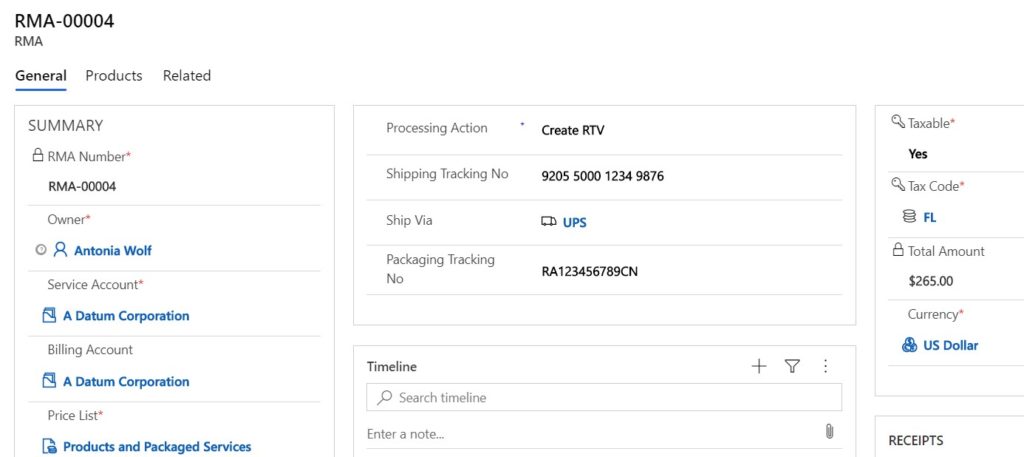
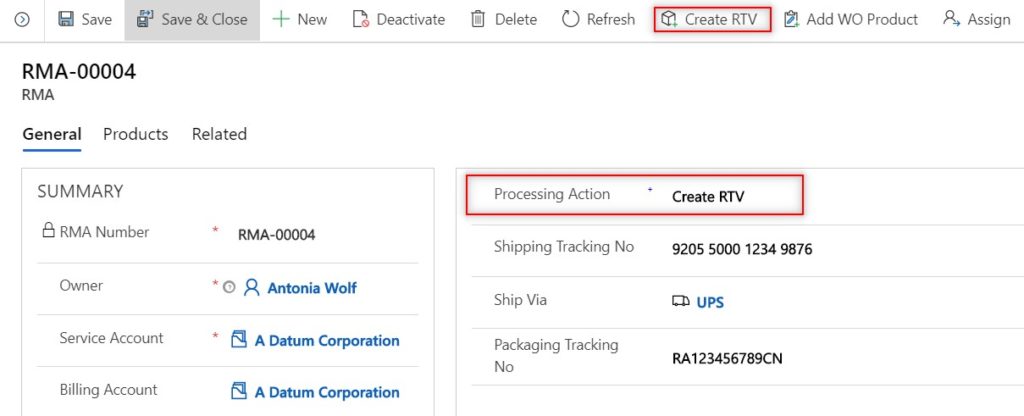
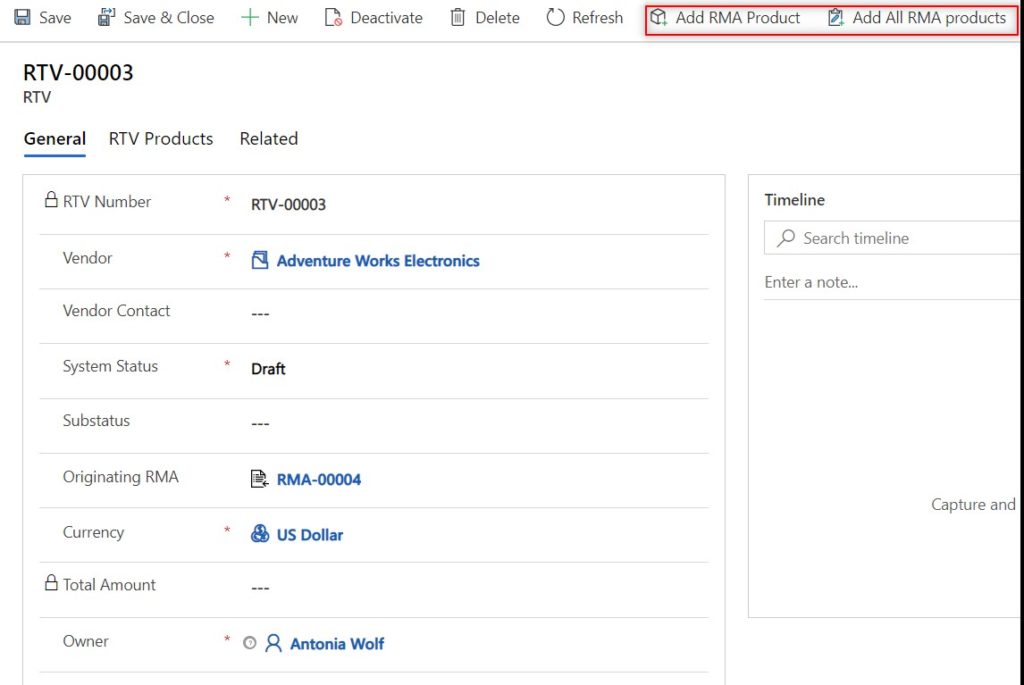
If it is decided that the returned products will be sent back to the medical OEM, you can easily create an RTV with the relevant RTV products from the RMA in one click. The RTV can be used as a reference to notate the returned items were sent back to the originating OEM, and how much credit to expect.
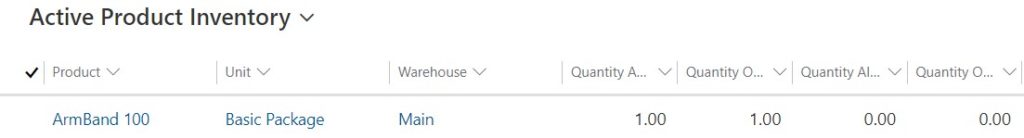
If a returned product is going to be sent back to a warehouse, you can specify the warehouse to return the part to. The on-hand product inventory will be updated automatically to include the quantities of returned items.
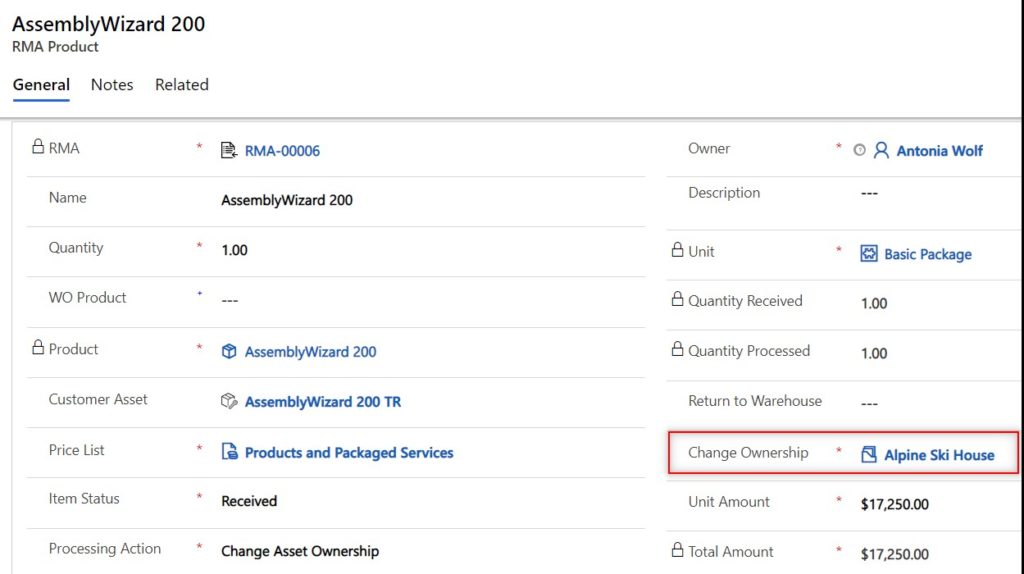
If a returned product is a medical device to be removed from a current customer location but needs to be tracked at a different customer location, use the change asset ownership to carry this return out. The entirety of the medical device history will still be tracked within the system. You will be able to see all work orders/maintenance that occurred on the machine at the previous customer locations.
All of the above will help your team record returns in real time, ensuring you keep track of inventory changes as well as any vendor credits you might be owed. Contact Velosio to see how we can help!